For Patients
Your Journey Starts Here
Hope backed by science. Explore active clinical trials designed to improve survival while maintaining quality of life.
Engineered viral platforms that activate the immune system to recognize and destroy cancer cells.
For too long, fighting cancer has meant harsh treatments and diminished quality of life. We believe in a different path.
Our mission is simple: to extend and improve lives through cancer immunotherapy innovation, without the toxicity of traditional approaches.
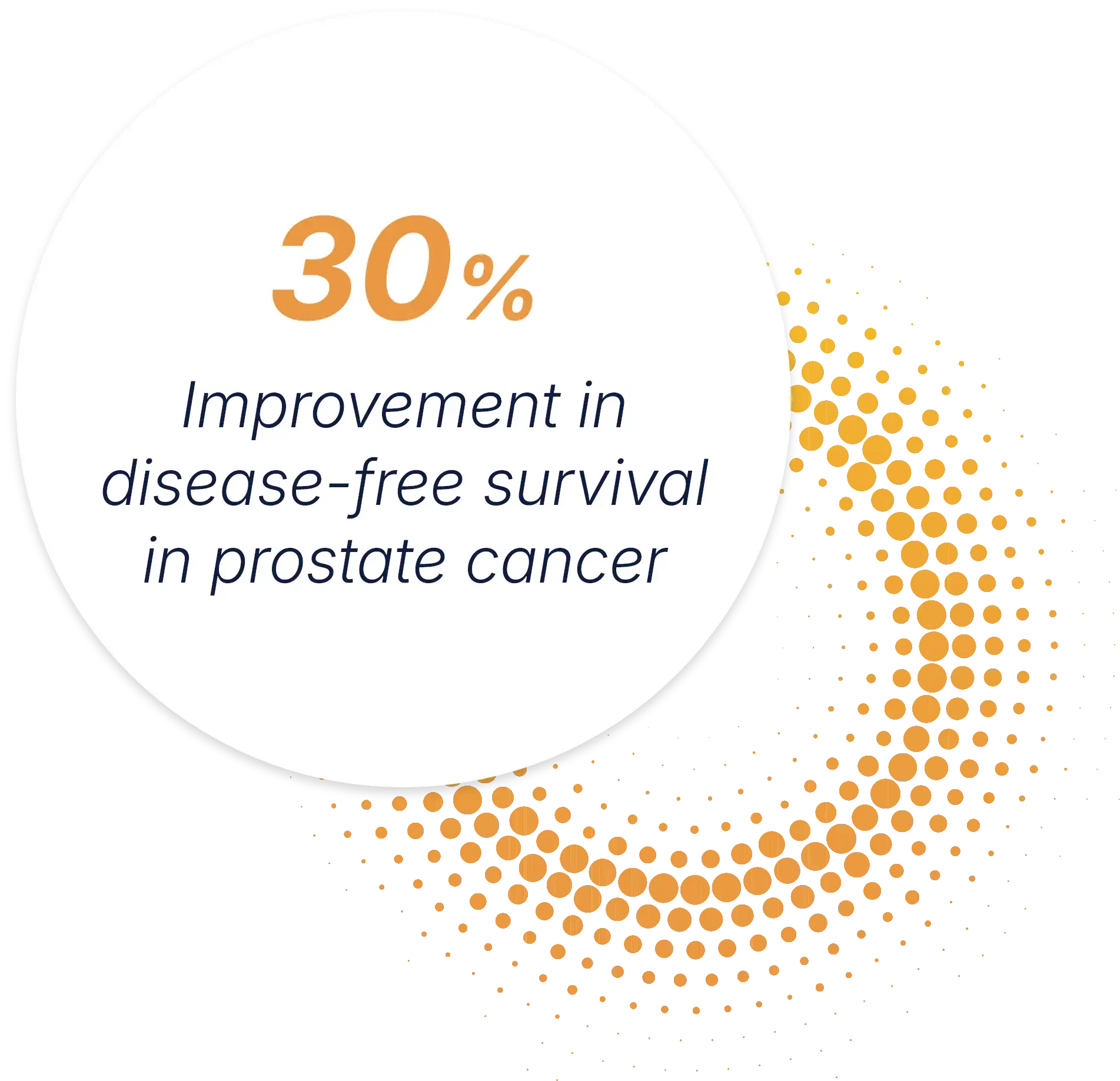
Phase 3 data for our lead therapy, aglatimagene besadenovec, is demonstrating meaningful outcomes for prostate cancer.

Candel has established two off-the-shelf clinical-stage investigational viral immunotherapies designed to deliver meaningful and durable clinical benefit through individualized immune activation based on novel, genetically modified viral constructs.
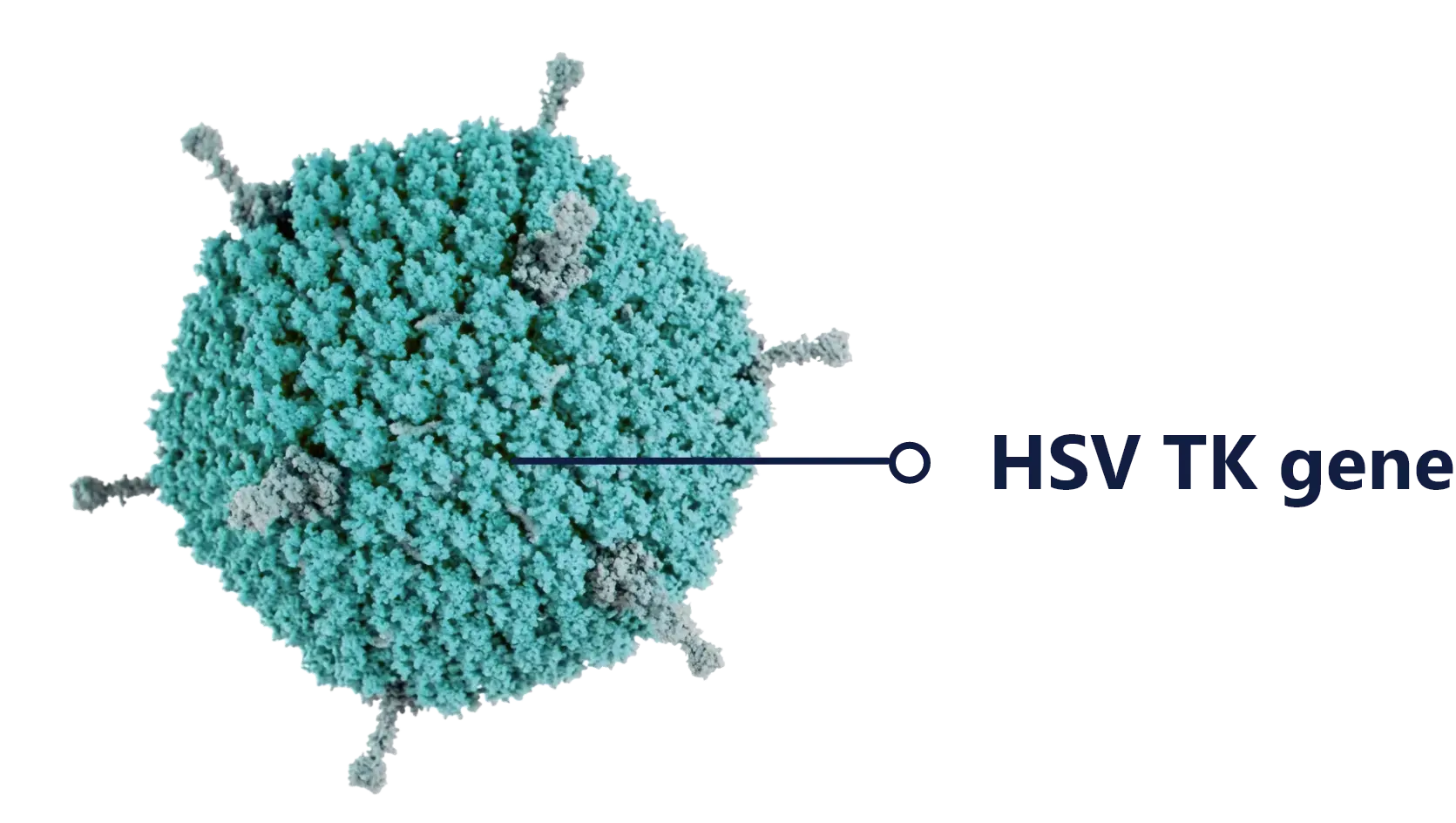
aglatimagene besodenovec (aglatimagene or CAN-2409)
.webp)
Aglatimagene is an adenoviral gene construct encoding HSV-thymidine kinase, injected directly into the tumor. When combined with FDA-approved oral prodrugs, it triggers immunogenic cell death and creates optimal conditions for CD8+ T cell–mediated anti-tumor immunity.
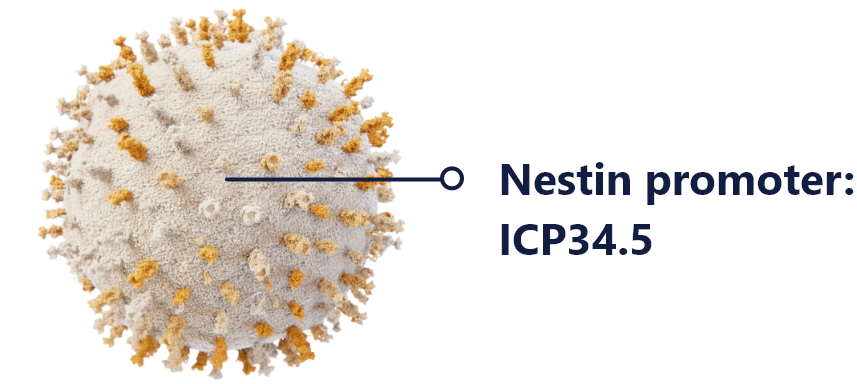
linoserpaturev or CAN-3110

Linoserpaturev is a replication-competent HSV engineered for selective killing of cancer cells while sparing healthy tissue. Controlled by the Nestin promoter, it targets high-grade glioma cells with precision. This tumor-specific approach enables multiple dosing without dose-limiting toxicity.
Whether you’re seeking treatment options, clinical evidence, or investment opportunities, we’re here to guide you.
For Patients
Hope backed by science. Explore active clinical trials designed to improve survival while maintaining quality of life.
For Providers
Evidence to inform your practice. Review Phase 3 data, safety profiles, and mechanism of action for viral immunotherapy across solid tumors.
For Investors
Clinical validation with commercial potential. Review Phase 3 data, BLA readiness, and our multimodal platform approach.